HealthTECH on Track to Double Program Impact
Growing Financial Award, Building Ecosystem Support and MATTER Co-Host Commitment Mark Expansion



Experience the process of driving innovation from idea to realization by participating in the Insight-led workshop entitled “How User-Centered Design Techniques Can Drive Actionable Innovation from Concept to Commercialization.”
 From medical device OEM founders to independent technology innovators, QMed magazine says that spying the workspaces of industry pros around the world reveals far more than simple lessons in neatness and clutter - it’s first line insight to the leading minds in medtech.
From medical device OEM founders to independent technology innovators, QMed magazine says that spying the workspaces of industry pros around the world reveals far more than simple lessons in neatness and clutter - it’s first line insight to the leading minds in medtech. The focused technology development efforts of Insight Accelerator Labs member PhysIQ were recently rewarded with FDA 510(k) clearance for the company’s personalized physiology analytics system. The startup’s technology, which has been categorized as a Class II device for patient monitoring by the FDA, analyzes a user’s physiological data - including heart rate, respiration rate, oximetry, and blood pressure - to create a personalized baseline for each user and identify exacerbations earlier.
The focused technology development efforts of Insight Accelerator Labs member PhysIQ were recently rewarded with FDA 510(k) clearance for the company’s personalized physiology analytics system. The startup’s technology, which has been categorized as a Class II device for patient monitoring by the FDA, analyzes a user’s physiological data - including heart rate, respiration rate, oximetry, and blood pressure - to create a personalized baseline for each user and identify exacerbations earlier.
“Without question, FDA clearance is one of the most significant hurdles to overcome, and we’re excited to see PhysIQ reach this major development milestone on their clear path to commercialization,” says Steve McPhilliamy, partner at Insight Product Development, and executive director of Insight Accelerator Labs.
PhysIQ is actively building its platform for any organization that is aggregating biometrics from different devices. The company has already partnered with Samsung on SAMI, a platform Samsung created to collect data from various devices, and with the Scripps Translational Science Institute (STSI) to create a program that uses health sensors to improve the health outcomes of Ebola patients. In 2014 the company raised $4.6 million in a round led by LionBird.
To learn more about Insight Accelerator Labs and its process for helping startups advance their technologies toward commercialization, contact Steve McPhilliamy at [email protected].
 Industry Leaders Select Five Up-and-Coming Innovators for WellTech Live Pitch
Industry Leaders Select Five Up-and-Coming Innovators for WellTech Live PitchIn the first round of the newly expanded Insight Product Development Startup Competition judging, professionals from MATTER, Baxter and Insight Product Development hand-selected the most promising early stage healthcare innovations to compete for $35K in cash and professional services to further advance their innovations toward commercialization. The finalists, which will live-pitch judges at WellTech on June 25th are 4D Healthware, Helix Analytix, Doc&i, Zest Health and TeleHealthRobotics.
Narrowed from a large round-one competition startup applicant pool, these five finalists will also join other competition front-runners in an innovation showcase at WellTech following the live pitches. Innovation showcase participants include Secarleye, UroProx LLC, Prana Diabetes, Janus Choice LLC, Neximatic, Varsa Health, and Integrative Pain Centers of America Inc.
To learn more about the Insight Product Development Startup Competition, or to register to attend the first phase of the competition at WellTech, visit: https://insightpd.com/healthtech-startup-competition/
 Insight director of research and strategy, Carolyn Rose, and senior researcher Abbe Kopra will reveal how high impact product design starts with a deep understanding of the needs of end users and customers at MATTER Chicago.
Insight director of research and strategy, Carolyn Rose, and senior researcher Abbe Kopra will reveal how high impact product design starts with a deep understanding of the needs of end users and customers at MATTER Chicago.
 As part of the Stakeholder Validation Program - a partnership between Insight Product Development and MATTER, Insight will lead multiple workshops to help startups identify the right stakeholders to approach, and the right questions to ask. MATTER member companies will learn how to further investigate their user hypotheses in an unbiased and informative way, assess whether the problem is painful enough that stakeholders would pay for a solution, confirm whether proposed solutions are seen as an effective “painkiller,” and emerge from the workshops with a template interview guide and a deeper understanding of the interviewing process.
As part of the Stakeholder Validation Program - a partnership between Insight Product Development and MATTER, Insight will lead multiple workshops to help startups identify the right stakeholders to approach, and the right questions to ask. MATTER member companies will learn how to further investigate their user hypotheses in an unbiased and informative way, assess whether the problem is painful enough that stakeholders would pay for a solution, confirm whether proposed solutions are seen as an effective “painkiller,” and emerge from the workshops with a template interview guide and a deeper understanding of the interviewing process.

Craig Scherer joined a panel of industry peers at the 2015 MD&M East Conference to share his professional insights. The panel discussion, entitled ‘From Design to Production: Turning a Great Design Concept into a Medical Device Masterpiece’ covered such topics as what makes a disruptive device a success, current devices that apply these disruptive innovation principles, who succeeds, fails, and why, and how to achieve the light bulb moment when designing your next device.
“It was a very lively discussion on the topic of creating disruptive innovation in the medical device industry,” said Scherer. “It was made more rich with panelists from consulting, academia and industry. The key masage? Know your users!”
For more information about this panel discussion, or for more information about the conference, visit the MD&M East Conference page.

With over 25 year experience innovating in the medical space, Craig Scherer, senior partner and co-founder of Insight was a judge for the medtech industry’s premier awards program, the Medical Design Excellence Awards.
The winners were announced at the MDEA ceremony held on June 9th in conjunction with the 2015 MD&M East Conference in New York, NY. The event celebrated the achievements of medical product manufacturers, their suppliers, the many people behind the scenes and was attended by over 500 medtech industry leaders.
To learn more about the mission, history and the award program overview, visit the MDEA Program Overview page.
With the potential to positively impact millions of ENT surgeries performed annually, the Olympus DIEGO … was awarded the silver award for Dental & ENT Surgical Tools in the 2015 Edison Awards program, Under the Science, Medical & Dental category. 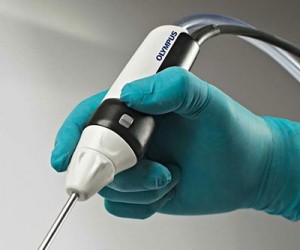 With its monopolar and bipolar energy blades, this innovation offers physicians an array of surgery options, reducing instrument exchanges during procedures, reduced bleeding and significantly shortened surgery times.
With its monopolar and bipolar energy blades, this innovation offers physicians an array of surgery options, reducing instrument exchanges during procedures, reduced bleeding and significantly shortened surgery times.
“We’re proud that our work has been recognized by this highly esteemed program,” says Craig Scherer, co-founder and senior partner at Insight. “The distinction of winning an Edison Award is significant, and underscores the value of our longstanding focus and approach to innovation.”
In the course this development project, Insight performed extensive contextual research to uncover unmet needs that resulted in the DIEGO® ELITE™ feature set and usability features to provide clear market differentiation, and developed the final commercial solution by combining and optimizing technologies that had never before been used in this configuration for this surgical procedure.
Read more about our work on the Olympus project and check out all 2015 Edison Award winners at http://bit.ly/1I3wlMW
